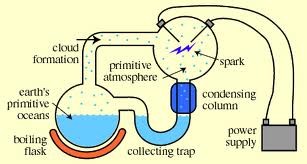
The focus of this experiment was to determine if amino acids could be produced by re-creating early Earth conditions. This experiment was done at the University of Chicago in 1953 by Stanley Miller. The experiment consisted of a boiling flask that was attached to a collecting trap that connected to a condensing column. This released steam that went to another bottling flask. Inside this flask were an anode and a cathode, which were connected to a power supply. This created an arc which was to simulate lightning from the early Earth’s atmosphere. The steam would condense and return to the original flask, where it was reheated and run through the cycle over and over again.
Harold Urey, Nobel laureate, was Miller’s doctoral advisor and Miller pulled heavily from his theories. Miller used a hydrogen rich formula that also included water ammonia and methane. The experiment was allowed to run for a week. Miller’s experiment showed that as much as 10 – 15% of the carbon was formed into organic compounds. Miller’s experiment showed that amino acids could be formed under conditions that could have existed on early earth.
Skepticism about this experiment has come about in recent years because of what some scientists believe the proto-atmosphere of earth was. Depending on what those gasses were, would radically change the outcome of the experiment. This hypothesis supports the idea that earth’s early atmosphere had very little hydrogen in it, that the hydrogen had boiled off into space. Some scientists believe that this early atmosphere would’ve consisted of carbon dioxide, water vapor and nitrogen. If you reproduce the experiment under these conditions you get cyanide and formaldehyde.
In 1995 Science magazine said that most experts now dismiss the experiment because “the early atmosphere looked nothing like the Miller Urey simulation”.
Another objection to this experiment was the continuous cycle that the elements were put through. Although it is believed that the early Earth did have frequent lightning storms, it would not have produced the amount of amino acids that the Miller experiment did.
It is challenging to locate knowledgeable persons on this topic, but you sound like you know what you are talking about! Thanks
michael kors purses
Can I just say what a relief to obtain a person who really knows what theyre talking about online. You certainly know the way to bring an concern to light and make it important. More individuals really need to read this and understand this side of the story. I cant think youre not far more common due to the fact you surely have the gift.
michaels kors handbags
Hello! I just would like to give an enormous thumbs up for the wonderful info you might have here on this post. I might be coming back to your blog for additional soon.
michael kors gansevoort